Cleanroom News, Contamination Control
Can Ethylene Oxide Ever Be Considered Safe?
Let’s face it, no-one is getting any younger. Even the fittest, most active and health-conscious among us will confront a health challenge at some point in our life and, as we age, those concerns tend to become more complex and require more careful treatment. Where youngsters can get away with the trauma of the occasional broken bone, the more – ahem – ‘mature’ members of the community face broader issues in dealing with time’s ravaging of the physical body. And these concerns can truly focus the mind. After all, dealing with the dual difficulties of health problems and insurance coverage battles is usually enough for most people to worry about. So it is understandable that few of us have the bandwidth to think of anything more than how to get better and how to pay for it. And we certainly do not want to have to worry about whether the equipment used in our care is fully sterilized.
But in the wake of the shuttering of two sterilization facilities in Michigan and Illinois, perhaps we should. Read on to learn more about how these developments could affect you…
One thing is certain in the medical field: contamination control is of paramount importance.
And this means sterilization of all materials – from the simplest wipes to the most complex replacement body parts. But what is the most efficient way of doing this? In terms of the devices and equipment used in patient care, cleaning and sterilization has traditionally been achieved in one of two main ways: steam processing in an autoclave or gamma irradiation. Let’s take a closer look at both options…
Perhaps the most simple method for sterilizing equipment is the autoclave, a device that essentially resembles and functions as an oversized pressure cooker.
With cylindrical chambers that withstand extreme pressure, the self-sealing steel vessel allows for the circulation of steam (or another gas) at a higher temperature than its usual boiling point. In terms of steam from water, the autoclave pressure causes it to be a full 20°C hotter, making it more efficient in terms of eliminating biological contaminants. And the process is relatively simple: place an object inside the chamber, seal, and then vacuum pump the air out (‘pre-vacuum’) or displace it by pumping steam in (‘gravity displacement’). Add more steam, but this time at higher pressure so that the temperature increases to between 121°C – 140°C (250°F-284°F), and let the object poach for between 3 and 20 minutes until sterilization has been achieved. Much like a domestic dishwasher, the efficiency of the process will depend upon the degree of contamination and the load size and positioning within the autoclave chamber.
As mentioned earlier, other gases may be used in place of steam when sterilization is not the aim of the process. For instance, autoclaves in industrial or scientific uses are employed to vulcanize rubber, nylon can be created from a concentrated salt solution via condensation polymerization, and circulating organic peroxides around ethylene in an autoclave creates polyethylene. But if sterilization is the aim of the game, steam from water is the best solution.
Unless, that is, gamma irradiation is your application of choice.
While images of Chernobyl, Three Mile Island, and Hiroshima may well spring to mind when we talk about radiation, gamma radiation through atomic decomposition is in fact a naturally occurring phenomenon and can be used very effectively in the sterilization, for instance, of biological materials for grafting. Allografts of connective tissue, bones, cartilage, tendons, corneas, and skin all benefit from gamma irradiation, as do instruments used in surgery such as scalpels, blades, hypodermic needles, and personal protective equipment such as gowns and gloves. In fact, between 40% and 50% of disposable medical products rely on this method of sterilization.(1)
And, at its core, gamma radiation is a relatively simple process.
Leveraging the fact that every atom or molecule releases electro-magnetic energy when it decomposes, the key to utilizing this energy release in service of sterilization is the selection of the correct emitter – more on this shortly. And surprisingly, it’s not exactly new technology. In fact, the first gamma irradiator built in the United States for this purpose was engineered in 1963. But how does controlled radiation result in effective sterilization? Via disruption! Gamma radiation is a type of short wavelength ionizing radiation that causes disruptions to a cell on the sub-atomic particle level. The radiation is produced when a substance termed the ‘emitter’ decomposes. The resulting drop in energy level causes the release of electro-magnetic radiation, which causes changes on the cellular level to the DNA or RNA of anything it encounters. In terms of the sterilization of medical equipment, the intended targets are, of course, any lurking microorganisms such as bacteria. And the power of the process is calibrated via the choice of emitter. A substance such as plutonium, for example, is a very effective sterilizer, but only if global destruction is the end goal. Cobalt-60, on the other hand, is the ‘Goldilocks’ solution – neither too weak nor too strong, but just right. And this is why it is custom engineered through the irradiation of Cobalt-59 to form the radioactive isotope by neutron capture. Correctly manufactured and stored, Cobalt-60 emits sufficient energy to kill microorganisms but not enough to cause treated items to become radioactive.
But, of course, both of these methods – radiation and the autoclave – have drawbacks…
On December 2nd, 2013, a truck carrying a teletherapy device used in cancer treatment was stolen near Mexico City, Mexico. The medical device was being transported from Tijuana to a disposal site and contained a rich source of Cobalt-60. The International Atomic Energy Agency (IAEA) classified this machine as a Category 1 source – the highest risk category that includes not only teletherapy sources but also irradiators and radioisotope thermoelectric generators (RTGs), the latter of which can also contain plutonium.(2) It is thought that the thieves were unaware that the machine, which they broke open, contained the radioactive substance which emitted 3000 curies of radiation. At a distance of 1 foot (12 inches) from the device, the perpetrators would have received a dose of 10 Rem per second – a typically lethal dose for half of a population given a 30-second exposure.(3) The opened equipment was ultimately recovered but not before alarm had sounded on the international stage. Then U.S. Press Secretary Jay Carney with the Obama administration issued a White House statement noting that the national security team had been watching the situation with a keen interest. ‘What’s most important is that the vehicle and equipment were recovered and the situation was resolved,’ he added with cool composure in a statement reported by The Washington Post.(4)
Although not as dangerous as the risk of radioactive contamination through misuse, dangers are nonetheless also posed by the autoclave process. Thermal degradation, for instance, can occur, causing damage to the surface of materials, and moisture-induced corrosion may occur over time causing the overall dimensions of objects to change. Given the precision of some of the tools concerned, clearly this is not an ideal scenario.
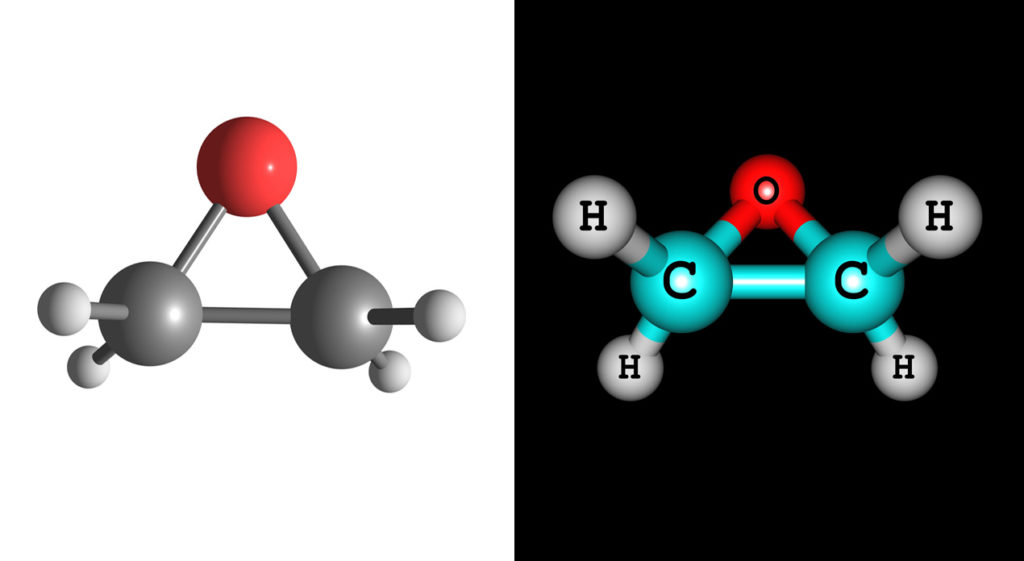
So if autoclaving is out of the question and the mere thought of wrangling radioactive materials leaves you in a cold sweat, we’re happy to report a third option: ethylene oxide penetration, better known as EtO.
According to a presentation by Dan Floyd of Nelson Laboratories, a provider of laboratory testing and advisory services to the pharmaceutical and medical industries, this sterilization method has gained broad acceptance and regulatory approval during its 60 to 70 years of use, with around 56% of medical devices currently being sterilized in this way – from wound dressings to stents and everything in between. And acceptance has been growing for several good reasons. Materials like plastics and polymers, which may not withstand high heats of the autoclave or be suitable for radiation sterilization, are ideal candidates for the low temperatures involved with EtO. In addition, this contamination control process is classed as ‘overkill,’ meaning there is a large safety factor built into the protocol. With radiation, the bio-burden is precision-calculated and in cases where it spikes, the sterilization effect may not be sufficiently strong. However, with ethylene oxide this is rarely the case given the large safety buffer, although the ‘overkill’ protocol does result in longer sterilization times. Moreover, EtO is a very small molecule, which means it can easily penetrate any object it sterilizes, even including entering into the walls of plastic devices.
However, there is a dark side to the use of ethylene oxide. As a gas, EtO is ‘an extremely flammable and reactive material [that] reacts violently with exposure to copper, copper alloys, and rust. [In addition it] is heavier than air and can travel long distances to a source of ignition and flash back to a leaking or open container.’(5) Moreover, it is classed as a genotoxic, mutagenic, and carcinogenic material due to its ability to ‘bind to nucleophilic biopolymers such as RNA, DNA, and proteins.’(6) The external signs of toxic exposure include, but are not limited to, eye irritation, lung damage, pulmonary edema, contact dermatitis, ataxia, headache, and nausea. Spontaneous miscarriage has also been reported in women working in situations of chronic exposure to the chemical.(7) To safeguard against exposure to EtO, it is recommended that appropriate personal protective equipment be used at all times. According to a fact sheet issued by the Environmental Health and Safety Department at Georgia Institute of Technology this includes ‘lab attire, safety glasses, lab coat and nitrile gloves […and] the only acceptable methods of respiratory protection against EtO are a military style gas mask […] or a Self Contained Breathing Apparatus (SCBA). [Furthermore an] air purifying respirator with an organic vapor cartridge is not sufficient to protect against EtO.’(8)
This is certainly sobering information. And this danger of toxicity is presumably why the Attorney General of Illinois last year filed a suit against commercial sterilizer Sterigenics U.S. LLC after the Environmental Protection Agency (EPA) alleged air pollution violations. Following a release of EtO at the company’s Willowbrook plant, Attorney General Lisa Madigan was dismayed to learn that a grassroots campaign had been lobbying against Sterigenics for at least six months, drawing attention to high levels of ethylene oxide around the plant. In an article published in the Daily Herald, Madigan is quoted as saying that ‘“For too long, Sterigenics emitted a dangerous, toxic chemical into the air putting the public’s health at risk.”’(9)
Unfortunately the Willowbrook incident was not the only one involving this gas. In Grand Rapids, Michigan, sterilizer Viant Medical announced a shuttering of its plant last month after air quality issues were reported to the Michigan Department of Environmental Quality (MDEQ). Viant Medical processes instruments such as catheters and surgical mesh and has a track record of air quality problems, having self-reported an emission from a faulty rubber seal back in July 2017. At this time, the downstream effects of this shutdown to the supply chain is unknown although the Food and Drug Administration (FDA) is recommending that manufacturers of affected devices consider contracting alternative sterilizers for future processing.
So with failures such as these in mind, and considering the potentially serious effects of EtO exposure, could there be a better way to ensure that surgical devices and supplies are correctly sterilized?
Perhaps so, if an initiative spearheaded by the FDA bears fruit. According to an article published last month in MD+DI, a resource for the Medical Device and Diagnostic Industry, the agency plans to announce a ‘public innovation challenge to encourage the development of new sterilization methods, which could include new devices or new modalities that are safe and effective for sterilizing medical devices.’(10) One of these contenders in this arena could be ozone sterilization, which so far has shown promise as an environmentally more sensitive method due to its decay back to oxygen. Leaving no residue, it has none of the moisture issues associated with the autoclave process and furthermore can be used on integrated circuits and polymers without the risk of radiation-induced corruption or materials degradation. Unlike Cobalt-60, it requires no transportation – and therefore presents no risk of theft or other alarming high jinx – and has been successfully used as a biocide and disinfectant in multiple applications.
We will watch the situation with great interest and are excited to see where this drive for innovation will lead. And, with a topic as significant as this one – with its broad implications for public health and safety both in and out of the medical setting – you can be sure that we’ll bring you further news and developments to keep you up to speed with this critical contamination control story. Thanks for reading!
Does the idea of Cobalt-60 leave you shuddering? Is ethylene oxide simply misunderstood? Could ozone be the future of medical sterilization processes? We’d love to know your thoughts!
References:
- https://tuttnauer.com/blog/sterilization-by-gamma-irradiation
- https://www-pub.iaea.org/MTCD/publications/PDF/Pub1227_web.pdf
- https://thebulletin.org/2013/12/lessons-from-a-mexican-theft/
- https://www.washingtonpost.com/world/stolen-cobalt-60-found-in-mexico-curious-thieves-likely-doomed/2013/12/05/262ef990-5d66-11e3-8d24-31c016b976b2_story.html
- https://www.ehs.gatech.edu/sites/default/files/ethylene_oxide.pdf
- ibid
- ibid
- https://www.ehs.gatech.edu/sites/default/files/ethylene_oxide.pdf
- https://www.dailyherald.com/news/20181030/government-lawsuit-alleges-air-pollution-violations-at-sterigenics
- https://www.mddionline.com/fda-scrambles-prevent-shortages-wake-sterilization-shutdowns



















HAVE AN IDEA FOR CONTENT?
We are always looking for ideas and topics to write about.
Contact Us