Bio-Technology, Cleanroom News
When Bacterial Biofilms Meet Magnetic Microbots
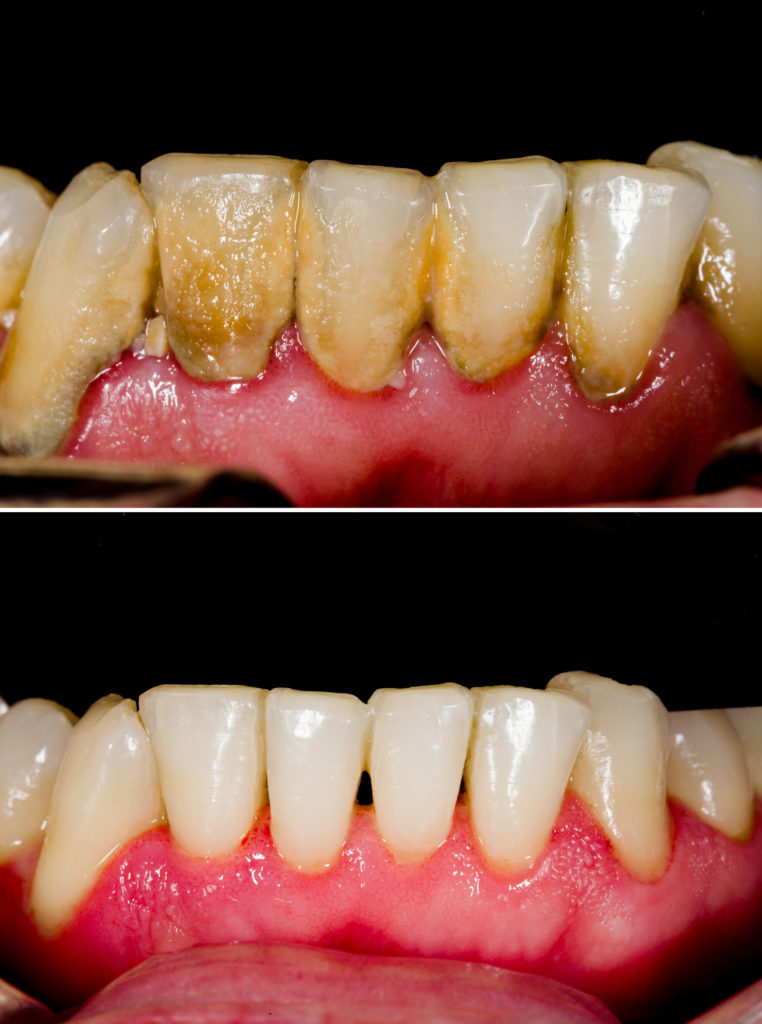
Can we have a show of hands for those whose oral health is such that they have never had a cavity, a crown, a root canal, or an extraction? Hmm….not seeing many hands going up right now. And that’s not surprising. Few of us are fortunate enough to avoid the excruciating anxiety of that long walk to the dentist’s chair. Of the terrifying words “You’ll just feel a little pinch…” as Novocaine slides into a tender gum. And the high pitched whine and smell of burning as a drill chews its way through enamel and into bone. And whether undergoing a root canal or a simple filling, one thought is universal to the experience:“Why?” Per the theory of Occam’s Razor, the best answer is usually the simplest one – and in this case it’s bacterial contamination.
But before rushing to judgement on oral hygiene, let’s map the common path we follow to the indignity of reclining in the dentist’s chair with a (literal) handful of latex-clad digits poking around our molars. The responsible parties, in this case, are the bacteria that colonize the mouth, forming communities on and between our teeth, and recycling sugars from our own food. And with feeding comes excreting, the results of which contribute to the formation of a biofilm called ‘plaque.’ Traditionally, we remove this detritus each time we brush and floss or have plaque scraped off at our twice-annual dental cleaning. But this is both a labor-intensive and seemingly low-tech approach in the fight against decay. Surely, in these days of extraterrestrial travel, cloning, and the bio-printing of human organs, there has to be a better way to keep mouths clean and free of biofilms? Good news: if current research coming out of the cleanrooms and laboratories of the University of Pennsylvania is a metric, perhaps there is. And if so, it involves legions – armies, in fact – of microscopic cleaning crews. Think that’s far fetched? Read on!
One of the hardest parts of maintaining excellent oral hygiene is the prevalence and reproductive speed of microorganisms in the naturally warm, moist, and bacteria-friendly human mouth.
Moreover, the problem is exacerbated by the fact that these organisms do not exist only in a planktonic state (independently floating in a solution) but also form cooperative collectives that have the advantage of strength in numbers. When bacteria band together to create living colonies, they develop a complex social system that is used both reproductively and defensively, and thrive on a sticky matrix that attaches to any surface. The matrix, or EPS, is a substrate for the bacteria, yeasts, fungi, algae, and protozoa, is composed of exopolysaccharides, proteins, and nucleic acids, and is strong enough to form an effective layer of protection for the bacterial colonies. Secretion of a glue-like substance allows the EPS to adhere to any surface and to perform as an external digestion system, with some varieties evolving with water channels that distribute nutrients throughout the colony. And biofilms can be equal opportunity hosts, becoming home to any mixture of bacteria. In fact, according to a publication of the University of Montana, ‘over 500 bacterial species have been identified in typical dental plaque biofilms.’(1)
Fortunately, the evolution of a biofilm is a multi-part process which gives us the opportunity to disrupt it at a variety of points.
During its five phases of development, the biofilm first moves from initial attachment, to permanent attachment before undergoing two phases of maturation. Finally, the process of dispersion begins – the spreading and colonization of additional surfaces and, in the case of dental plaque, the eventual visit to the dentist. But let’s not get ahead of ourselves – the processes of initial and subsequent attachments are critical to understanding the mechanisms at work in bacteriological contamination control. So let’s return to the very start…
Following the formation of a film, hydrophobic effects and van der Waals forces – those same forces we discussed in regards to gecko toes in space – adhere the matrix to a surface. Secondary, more permanent bonding is achieved via cell adhesion techniques using the motile microbes’ pili – tiny hair-like surface appendages – with non-motile bacteria adhering directly to the scaffold or to earlier bacterial colonists. And this is where the development of biofilms becomes especially interesting. During the growth of the colony on a surface, bacteria use quorum sensing in order to produce the best environment for reproduction. Quorum sensing is the ability to detect population density – how big a colony is growing – and to control it through gene regulation. First identified in the early 1970s, quorum sensing is utilized by bacteria to control specific phenotype expressions which in turn coordinates behaviors. And, depending on the bacterium – gram-positive or gram-negative – this is either a bi-partite or a tri-partite process.
Let’s use the example of a gram-negative bacterium. Initially, a signaling molecule – an autoinducer – must be secreted. In the case of oral biofilms, this signaling molecule could be N-acyl homoserine lactone (AHL) which is thought to change the surface tension sufficiently to disrupt Marangoni flows, thereby enhancing colony motility and allowing it to swarm.(2) Once secreted, the AHL binds directly to transcription factors – proteins that regulate genetic switching (the ‘turning on’ and ‘turning off’ of genes) – to ensure their correct and timely expression for the duration of a cell’s lifetime. In gram-negative bacteria a different signaling molecule – autoinducing peptide, or AIP – is secreted which must first bind to a receptor and activate kinase – an enzyme critical in cellular transport, protein regulation, and metabolism among other functions – before a transcription factor is produced. Once the autoinducer is bound to the transcription factor the process of creating a biofilm begins and, depending upon type and location of the film, it can range in density from just a few molecules to several inches in thickness.
Interestingly, given that they can form on any non-shedding surface, biofilms are relatively ubiquitous and different types can develop and thrive in even the most extreme environments.
Moreover, in certain circumstances they are beneficial to their environment, existing symbiotically with plants, for example, or contributing to the broader eco-system, as in rivers or streams. But when they not only do not contribute to their environment, but actually cause harm, they need to be removed. And quickly. In terms of the oral biofilms, until now the deft application of a toothbrush, a fluoride-based paste, and a swish of antibacterial mouthwash has been the best we can achieve, at least on a daily basis. But fortunately technology is on our side in the war on harmful microbes as an article recently published in Cleanroom Technology reveals. In ‘Princeton researchers discover new biofilm removal technique’ Sophie Bullimore examines how capillary peeling focuses less on biological resources and more on leveraging mechanical engineering to cleanly and completely remove biofilms. Quoting a paper published in Advanced Materials, a peer-reviewed scientific journal covering materials science, Bullimore notes comments made by study co-author Bonnie Bassler: ‘“By investigating and defining the material properties of bacterial biofilms, rather than their biological properties, we have invented a new method for detaching entire biofilms.”’(3) And Bassler, the Squibb Professor of Molecular Biology at Princeton and Howard Hughes Medical Institute Investigator, came to this method seemingly via reverse engineering the opposite problem. How? Let’s dive a bit deeper…
Biofilms essentially exhibit many of the same characteristics as hydrogels – both are composed of around 90% water and have a structure that allows them to be very flexible, elastic, and viscous. Hydrogels are frequently used in the medical field as wound dressings and as a vector for drug delivery but they are notoriously fragile as the very structures that make them stretch also cause them to fragment under excess force. In other words, they have to be handled with extreme care. Using cleanroom technologies, researchers at Princeton laboratories recognized the limits of the physical manipulation of hydrogels and zeroed in on the attachment mechanisms of V. cholerae biofilms in order to better understand how to avoid fragmentation in beneficial hydrogels. And that is when the discovery was made. In examining fragmented biofilm patches, the team saw that while the edges of the patch were hydrophobic, the surfaces to which they were attached were hydrophilic – a clear point of tension. By introducing water into the space between the biofilm and the surface, researchers were able to slowly lift the biofilm from the surface without it breaking apart. Think of it like the process of carefully peeling away a sales sticker from the cover of a new book. And for applications where the contact surface is already in an aqueous environment, it is thought that the introduction of air bubbles between the surface and the biofilm may be equally as effective.
Biofilms, in all their multitude of guises, are a serious concern in a variety of industries – from food processing to environmental contamination control, healthcare to optics, pharmaceuticals to aerospace.
All of this seems well and good but the question necessarily arises as to the applications for this technology. With a genesis in dentistry, is it useful outside of the oral healthcare field? Absolutely! Biofilms, in all their multitude of guises, are a serious concern in a variety of industries – from food processing to environmental contamination control, healthcare to optics, pharmaceuticals to aerospace. In fact, given that they can attach to almost anything, contamination is theoretically possible in the manufacture or processing of any object that has a surface. Moreover, if not eradicated from the manufacturing cycle, initial microbial contamination can not only impact multiple batches of products but also lead to the development of resistant biofilms that are even more difficult to remove successfully. Which is why a new label claim announced by STERIS Corporation, with global headquarters in Mentor, OH, is particularly exciting in terms of contamination control. Already labelled as a disinfectant and virucide, STERIS ProKlenz® ONE Alkaline Cleaner is now recognized as a remover of ‘one of the most challenging soils in the pharmaceutical and biopharmaceutical industries.’(4) Developed for use on non-porous surfaces, the product is low-foaming and aimed at clean-in-place (CIP) and clean-out-of-place (COP) applications in the pharmaceutical, cosmetic, biotech, and nutraceutical industries. Strongly alkaline, it acts on process residues ‘from fermentation by-products to silicone-based emulsions and lubricants’ and is accredited as a validatable cleaner.(5)
Clearly this new label claim is welcome news for surface contamination removal, but it is not the only exciting development. According to an article published last month in Science Robotics, researchers at the University of Pennsylvania have gone a step further by engineering an army of cleaner robots, controlled via magnets. Pioneered by Hyun (Michel) Koo of the university’s School of Dental Medicine in collaboration with Edward Steager from the School of Engineering and Applied Science’s General Robotics, Automation, Sensing, and Perception Laboratory (GRASP), two platforms were created to degrade and remove dental biofilms. Termed catalytic antimicrobial robots – or CARs – the micro robots were built with iron-oxide nanoparticles and can be either suspended in a solution or embedded into gels. In combination with medical imaging to detect areas of significant growth, the micro robots are surprisingly precise in removing film via a plowing or shattering motion across surfaces or within tubes. In fact, one of the potentially most useful applications in the field of oral health would be the cleaning of the isthmus, the narrow channel between root canals which is notoriously difficult to access using traditional techniques.
The eradication of harmful microorganisms – whether planktonic cells or biofilms – can be as simple as using a recommended cleaner and wipes to decontaminate a cleanroom surface, or it can be as complex as deploying magnetically controlled micro robots in a catheter or a root canal.
How exactly this technology will look in its future clinical application is currently unknown but the work of Koo’s team certainly seems promising. So much so, in fact, that it has received support from many agencies, including the Penn Center for Health, Devices, and Technology, the National Institute for Dental and Craniofacial Research, and the National Science Foundation. And the technology also underscores an important point in terms of general contamination control: variability. The eradication of harmful microorganisms – whether planktonic cells or biofilms – can be as simple as using a recommended cleaner and wipes to decontaminate a cleanroom surface, or it can be as complex as deploying magnetically controlled micro robots in a catheter or a root canal. And assuming the correct selection is made and procedures are followed, no single method is inherently superior to another. They are all necessary and vital tools in the on-going and ever evolving fight against contamination. A fight that neither side will ever definitely win.
The intersection of biomedical science and mechanical engineering represents an exciting area of research. We’d love to know your thoughts!
References:
- https://www.cs.montana.edu/webworks/projects/stevesbook/contents/chapters/chapter001/section002/green/page001.html
- See https://www.pnas.org/content/pnas/103/40/14965.full.pdf
- https://www.cleanroomtechnology.com/news/article_page/Princeton_researchers_discover_new_biofilm_removal_technique/151855
- https://www.sterislifesciences.com/news-and-events/news/2018/december/steris-corporation-announces-addition-of-biofilm-removal-claim-using-proklenz-one
- ibid



















HAVE AN IDEA FOR CONTENT?
We are always looking for ideas and topics to write about.
Contact Us