Cleanroom News, FDA
Silicone Contamination and the FDA
Opening soon in theaters across the nation, the movie Wonder, adapted from the eponymous bestseller by R. J. Palacio, is already the subject of intense controversy. The story centers on the character of August Pullman – ‘Auggie’ to his friends – a ten year old boy, about to start school. And Auggie is no ‘normal’ 5th grader: he suffers from a condition which, in the book, is termed mandibulofacial dysostosis but which may actually be Treacher Collins Syndrome. TCS is a genetic condition caused by mutations of the EFTUD2 gene, which lead to the abnormal development of facial bones and tissues. An unlikely hero, Auggie’s cinematic narrative takes the viewer on an extraordinary journey of prejudice based in fear of ‘otherness’, through compassion, and onto acceptance, ultimately imparting the message that, as online movie portal Rotten Tomatoes suggests, ‘you can’t blend in when you were born to stand out.’(1)
Although Wonder has provoked some measure of backlash from critics within the craniofacial-conditions community, other commentators have praised the film for shining a light on a condition which, according to the National Organization for Rare Diseases (NORD), is suffered by an estimated 1 in 10,000 to 1 in 50,000 individuals.(2) A type of facial dysmorphology, Treacher Collins Syndrome results in missing or abnormally formed ears (sometimes accompanied by hearing loss and the potential for speech problems), eyelid abnormalities (often affecting vision), sunken cheek bones, stunted growth of the chin and mouth, cleft palate, and under-developed jaws. Some 5% of sufferers also exhibit developmental or neurological delays.
For most Treacher Collins patients, cosmetic surgery starts early with infants enduring their first procedure between the ages of 12 to 24 months. The character Auggie says he’s had 27 different procedures, confiding wryly to a friend that ‘“This is after plastic surgery. It takes a lot of work to look this good.”’(3) And as they develop and enter puberty, individuals like Auggie require additional surgeries to adjust to the changes in their bodies.
In our looks-obsessed society, for facial dysmorphology patients cosmetic procedures mean the difference between at least partial acceptance and outright rejection.
They are medically necessary on one level to address issues such as respiratory problems associated with choanal atresia (blockage of the nasal passages), congenital heart defects, and hearing impairments due to abnormalities in the ear canal, inner ear, and ear bone formation.(4) But they also play a significant role in helping the patient face an increasingly body-obsessed world.
But what about the other face of cosmetic surgery? What of the growth industry that has sprung up around a variety of body insecurities, often of a psychological rather than physical nature? What about our quest for physical perfection and beauty through elective cosmetic surgery, aka plastic surgery?
In the celebrity world, few proponents of cosmetic procedures are as well known as the seemingly ubiquitous Kardashian clan. Famous for being…well, famous really, Kim Kardashian – or rather a certain portion of Kim Kardashian – hits the headlines with monotonous regularity due to the bodily manipulations in which she indulges. What started as an apparent attempt to rectify an imperfection caused by a cortisone shot for psoriasis has led the reality TV star to acknowledge her use of silicone implants to ‘enhance’ the shape of her derrière. But although Hollywood would have us believe that these implants are a common procedure in the cosmetic surgery field – almost a rite of passage – they are by no means devoid of risk.
Presenting herself as a medical professional from a plastic surgeon’s office, Cornelius administered silicone shots to the teen who later experienced breathing difficulties and subsequently died.
In North Carolina earlier this year, Kayvonceya Iman Cornelius was charged with the second-degree murder of a teen who was transitioning from male to female. Presenting herself as a medical professional from a plastic surgeon’s office, Cornelius administered silicone shots to the teen who later experienced breathing difficulties and subsequently died. Cornelius also treated another patient with about-to-expire silicone said to have originated in the plastic surgeon’s office, although he did survive the experience. Hospitalized with breathing difficulties, the poorly administered silicone was shown to have migrated through his body with some settling in his lungs. He is now permanently partially disabled and reliant on pain medication to manage his condition.(5)
And, while disturbing, these are not the only cases of botched cosmetic procedures or those involving sub-standard products to have come to light. According to dermatologist Amy Newberger, even correctly administered, medical-grade silicone can harden over time and become more visible as skin ages and thins. Citing a patient who’d received injections in her 40s to fill in developing wrinkles, Newberger noted that by her 60s the patient had started to look ‘“like a ‘klingon’ [where the…] permanently raised ridges of silicone became discolored.”’(6)
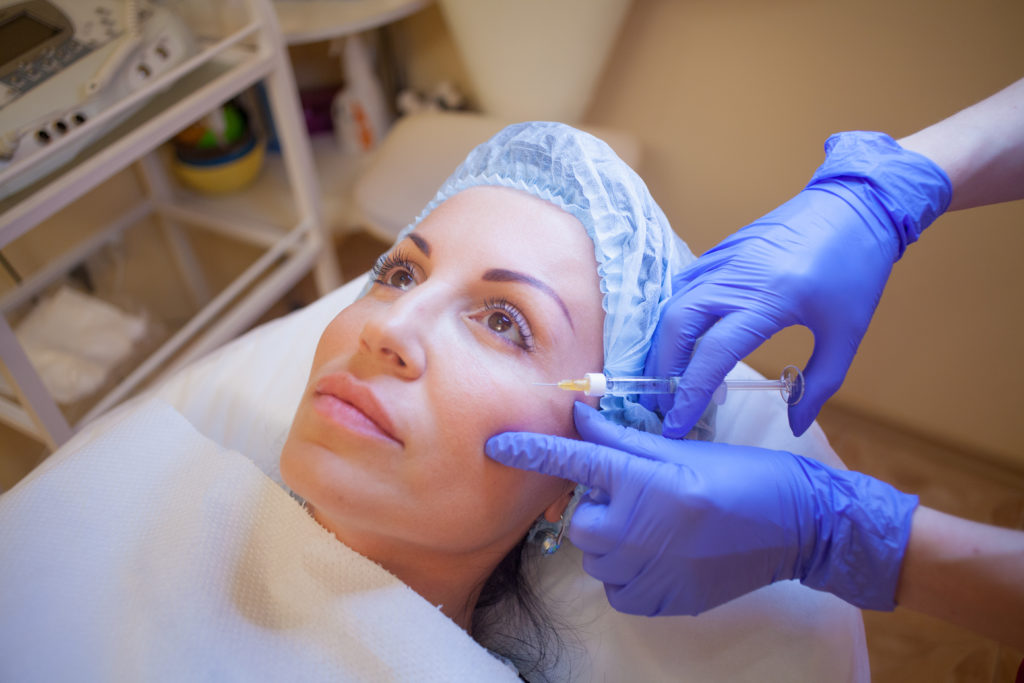
And although deeply unfortunate for the individual concerned, this is perhaps one of the less dangerous cases. According to an article by National Public Radio (NPR), the administration of injectable silicone is a thriving underground business, targeting those driven by a need for rapid physical change.(7) Alongside middle-aged women with a developing phobia for their own reflection, transgender adolescents are also a vulnerable population insofar as they form the perfect combination of desperation, the perceived invulnerability of youth, and teen impatience. Recommended treatment for those wishing to transition can take years and adolescents are not exactly known for their long-termism making them ripe for events such as ‘pumping parties’ in which potentially unlicensed ‘cosmetologists’ administer often questionable silicone injections to create and plump out curves.
In addition to the potential lack of licensing there is also the question of the purity – or otherwise – of the silicone.
Medical grade silicone is approved by the Food and Drug Administration (FDA) only for use in breast augmentation/reconstruction or ocular surgery. In the latter, the substance is used to create a matrix that supports a detached retina in place until it attaches back to the inner surface of the eye. Usually the silicone scaffold is removed within 12 months of the initial procedure.
In non-medical situations, however, not only might the procedure be compromised by the skills – or otherwise – of the practitioner, but also by the materials used. Quoted in NPR’s article, ‘Silicone Injections May Harm Some Patients,’ Dr. Nick Gorton of the Lyon-Martin Health Services clinic in San Francisco notes that ‘“there is no way to verify if they’re using medical grade silicone. You can go to hardware stores and buy a big tub of it […] The element is the same, but it’s just not the same safety or purity or quality.”’(8) And the effects of contaminated silicone are myriad: from severe infection to burns to embolism to a sloughing off of the skin due an adverse reaction. And of course, there’s the possibility of death.
So let’s consider specifically what the FDA perceives to be the most significant potential dangers of silicone implants.
In addition to migrating and hardening over time, implants can also cause capsular contracture (fibrous tissue growth around the implant causing hardening and/or distortion of the area), breakage and/or microleakage can occur, calcification of the fibrous capsule is possible, the potential risk for autoimmune diseases such as lupus is increased, and the incidence of cancer also rises in the presence of silicone implants.(9) Some of the disease risk is associated not so much with the silicone itself but with the coatings applied to the prosthetics. Way back in the early 1990s an FDA internal memo noted that the polyurethane foam covering of a popular implant from Bristol-Myers Squibb, the Même, degraded within the body to form 2,4 toluenediamine (TDA), a known carcinogen in animal studies. Estimating the risk elevation for cancer due to the presence of TDA, the FDA suggested an uptick from 6 in 1 million to 130-180 in 1 million. Clearly a substantial rise.
So if the substance is not inherently safe and if the underground practitioners are not necessarily of the highest medical caliber, how can the industry be regulated and controlled?
While some in the medical community have accused the FDA of being slow to act on the issue of silicone implants and prostheses, the agency does produce a useful factsheet, ‘Check Before You Inject.’(10) Stressing that any procedure should be carried out by a licensed healthcare provider, the document emphasizes the importance of becoming an informed patient – knowing what is approved or otherwise, and understanding the fillers and situations to avoid. The potential medical complications are also clearly laid out and the risk of purchasing fake, tainted, or contaminated fillers online is also stressed. Overall it’s a useful document for those prospective patients with the insight into their long-term health to absorb the information, but one that may be easily ignored by others.
And although there is a limit to how governmental oversight can shape social attitudes to issues like this sort of body modification and sculpting, one area in which it can act is the regulation of commercially available products used on the legitimate market. Take, for instance, devices manufactured by Irish pharmaceutical company, Allergan.(11)
And at the manufacturing level, Allergan utilizes both aseptic manufacturing and terminal sterilization processes, and adheres to the most stringent standards from the different countries they supply.
With a strong record for product safety, Allergan manufactures a spectrum of pharmaceutical products including drugs, biologics, and medical devices such as breast implants and intra-dermal injectables. And in the case of these devices, products are subject to additional quality assurance verifications before entering the multi-national supply chain. Using a ‘proof of process’ compliance system, each device is verified according to market-specific regulations, conforming to each country’s regulations on safety and purity. Device performance is monitored over the duration of its lifetime, and the company works closely with the FDA and the FBI to combat counterfeiting of its products. As a meta-picture, patient safety seems to be at the forefront of Allergan’s systems, with the company adhering to Good Clinical Practices (GCP) and the Declaration of Helsinki – the World Medical Association’s (WMA) cornerstone position paper of human research ethics. And at the manufacturing level, Allergan utilizes both aseptic manufacturing and terminal sterilization processes, and adheres to the most stringent standards from the different countries they supply.
In the final analysis, our short-term quest for physical perfection – especially for facial beauty – may often outweigh our long-term desire for health. It’s an unfortunate fact of human nature. But as with most subjects, knowledge truly is power. Knowing which substances, providers, and techniques to avoid is critical in keeping contaminated products out of our bodies and unlicensed or shady ‘practitioners’ out of business. No breast augmentation or facial wrinkle is worth running the risk of introducing potential carcinogens into our systems or suffering a device breakdown that results in pulmonary embolism. It’s perhaps time to rethink our notions of beauty…
Do you have experience with cosmetic surgery? Under what circumstances would you consider silicone implants? We’d love to know your thoughts!
References:
- https://www.rottentomatoes.com/m/wonder/
- https://rarediseases.org/rare-diseases/treacher-collins-syndrome/
- http://www.refinery29.com/2017/11/173462/auggie-from-wonder-disease-treacher-collins-syndrome
- https://rarediseases.info.nih.gov/diseases/10056/mandibulofacial-dysostosis-with-microcephaly
- http://www.wral.com/virginia-man-says-he-almost-died-from-backroom-silicone-injections/16604297/
- https://www.npr.org/templates/story/story.php?storyId=104724677
- ibid
- ibid
- http://www.breastimplantinfo.org/what-you-need-to-know/aathe-fda-breast-implants/the-fdas-regulation-of-silicone-breast-implants/
- https://www.fda.gov/downloads/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/UCM584838.pdf?utm_campaign=FDA%20Warns%20Against%20Use%20of%20Injectable%20Silicone%20for%20Body%20Contouring%20and%20Enhancement&utm_medium=email&utm_source=Eloqua&elqTrackId=3AF82AF1437B074A0230626444A91BD7&elq=0083d14d82134dd687f9462772b10614&elqaid=1318&elqat=1&elqCampaignId=810
- In the interests of full disclosure, Allergan is a customer of Berkshire Corporation.



















HAVE AN IDEA FOR CONTENT?
We are always looking for ideas and topics to write about.
Contact Us