Cleanroom News, Cleanrooms, USP 797
Compounding Pharmacies: Exploring the Insanitary/Unsanitary Conundrum
Introduction
When it comes to pharmaceutical products, medications, or nutritional supplements, as members of the contamination control industry – and as consumers – we like to believe that they are health-promoting, safe, and pure. From a professional standpoint, we understand the critical importance of preparing, packaging, and storing materials and compounds that will be absorbed into our bodies in sanitary, sterile, and hygienic conditions. And we expect others to understand and respect this too.
So the publication last month of a draft document ‘Insanitary Conditions at Compounding Pharmacies: Guidance for Industry’ by the Food and Drug Administration (FDA) has us scratching our heads once again.(1) On one hand, we welcome governmental oversight of an at times problematic industry. But equally, we can’t help but reflect with some measure of consternation on the continuing need for this document. Could it really be that the compounding pharmacy industry needs to be taught how to operate in a sanitary way? Given the number of articles we’ve published on Cleanroom-News.com on pharmacies whose practices have ranged from flouting conventions to an outright breaking of the law it might indeed appear so.
From the FDA’s standpoint, the rules – while complex and numerous – are clear. To mitigate the risk to public health, all pharmaceuticals must be produced under certain conditions and within stringently controlled environments. The conditions for manufacturing are laid down within the Federal Food, Drug, & Cosmetic Act (FD&C Act) which came into effect in 1938. Authored by then U.S. Senator from New York, Royal S. Copeland, this new set of laws replaced the ‘Pure Food and Drug Act’ of 1906 which had failed to prevent the deaths, in 1937, or some one hundred victims of ‘Elixir Sulfanilamide,’ a preparation that combined sulfanilamide – an antibiotic and antibacterial – with the highly poisonous solvent diethylene glycol. Following a public outcry over the deaths and the suicide of the lead pharmacist while awaiting trial, the FD&C Act with its ten separate chapters and myriad subsections aimed to toughen up the law and prevent history from repeating itself. But as recent cases have shown, there’s still a long road ahead.
Although Section 503A of the Act specifically covers pharmacy compounding it might appear that modern compounders are not necessarily up to speed with the regulations outlined. Which may, of course, be the reason for the FDA’s August release of the non-binding recommendations on questions of insanitary conditions. So before we delve into that document, let’s take a moment to review the definitions of terms involved…
The Insanitary/Unsanitary Conundrum
Insanitary. Unsanitary. Do you know the difference? Well, you don’t have to be a linguistics major to know that one is not the reverse of the other. Entering the English language in the early 1870s, the term ‘unsanitary’ connotes a state of unhealthiness, of dirt, one that is conducive to harboring or spreading disease. Conditions, places, buildings, physical environments may all be ‘unsanitary,’ as may practices, procedures, or operations. Insanitary, which appeared in the lexicon around the same time, simply implies a lack of sanitation.(2) A slight but, for our purposes, significant difference – and one fully recognized by the FDA.
Within the contamination control industry, examples of insanitary conditions may occasionally, albeit hopefully infrequently, conjure up images of visible filth. We have, for instance, reported on such conditions in a seafood processing plant in Maine where samples of rodent feces ‘too numerous to count’ were observed in an area where equipment was cleaned and black mold clung to a walk-in freezer.(3) But usually, we’re not necessarily talking about that level of contamination. As in the multiple cases we’ve recently cited of troubled compounding pharmacies, we’re more concerned with the unseen contamination that lax oversight, inadequate operator training, or simple lack of SOPs allows into protected areas. (4)
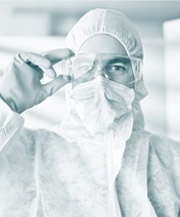
Examples of insanitary conditions, as cited by the FDA, include (but are not limited to) microbial contamination by molds or bacteria, environmental contamination (rust, human hair etc), cross-contamination during handling, inadequate ventilation and air flow sanitation. Although all of these conditions are applicable to the production of both sterile and non-sterile drugs, it is within the sterile environment that the FDA lays out its specific recommendations. Listing out aseptic practices that risk introducing contamination into a cleanroom environment, the draft document highlights no fewer than thirteen ways in which sterility can be compromised. An inspector witnessing any of the referenced infractions – from a technician’s bare hand contact with the outside of a glove, to improper gowning, to using non-sterile tools or disrupting a unidirectional airflow by moving too quickly – will quickly result in a warning letter from the FDA. Or, in the case of one of the most egregious of perpetrators, of far worse.
Insanitary Conditions…on Steroids!
In 2012, an epidemic of fungal meningitis gripped the nation as more than 800 people were sickened. The cause of this uncommon outbreak was traced back to supplies of an injectable steroid, methylprednisolone acetate, commonly used for pain management. Produced by a compounding pharmacy, New England Compounding Center (NECC), whose president and head pharmacist, Barry Cadden, had done more than push a few boundaries, this drug was contaminated with fungal spores that found their way directly into unsuspecting patients. Of those who received treatment from this tainted batch of product, 64 died in 20 separate states.
As we reported in an earlier article, the FDA’s subsequent investigation unearthed a shocking litany of failure, ingrained bad practices, conspiracy, and corporate malfeasance. Highest on the list of problems was the state of the company’s ‘cleanroom,’ in which a leaky boiler oozed a pool of stagnating water, uncleaned powder hoods grew a mossy skin of dirt and fuzz, and the main air intake vented within 30 yards of a contaminant-spewing recycling plant. This was the environment in which drugs that were to be infected directly into some patients’ spines were autoclaved for only 75% of the required time, in order – the FDA alleged – to squeeze in production of an extra two batches per day.(5)
In the wake of the horrendous death toll, NECC’s executives and pharmacists faced criminal prosecution on charges ranging from violating federal drug laws, to racketeering, to fraud and conspiracy, and eventually to murder. Glenn Chin, the supervising pharmacist, was arrested at Boston’s Logan Airport while attempting to board a flight to Hong Kong, a country that since its transfer of control from the United Kingdom to China no longer has a clear-cut extradition policy with the US. NECC’s president, Barry Cadden, didn’t even bother with the drive to the airport: knowing he was subject to arrest, he simply waited at home for federal agents to arrive. Both suspects are still awaiting their day in court, with Cadden’s trial scheduled for January 2017.(6) If convicted, both men could face life in prison.
Don’t Be Like Cadden.
Although the NECC’s case is unique in many ways – the fraudulent use of bogus patient name lists and acting as a manufacturer instead of a pharmacy by selling large quantities of drugs without specific prescriptions – the problems within the cleanroom are sadly not. But they are ultimately avoidable. All it takes is targeted operator training backed up by a strong framework of up-to-date standard operating procedures. Let’s get back to basics…
Cleaning and disinfecting. It’s a fact that humans are the #1 source of contamination within a cleanroom environment. Why? Because, even when almost motionless, each member of your team will generate approximately 100,000 particles per minute. And when you adjust that figure to account for movement, the estimate increases by a factor of 50! So constant cleaning is vital within critical environments. But using the correct cleaning supplies within a contamination-controlled area is also vital – using non-sterile disinfecting agents (fluids, wipers, swabs, or pads) in an aseptic area could do more harm than good, spreading microbial contamination more widely.
In any ISO Class 5 or Primary Engineering Control (PEC) areas, validated sterile wipes are recommended. (For a refresher on what ‘validated sterile’ means for your organization, please review our article.) Berkshire produces several lines of gamma-irradiated validated-sterile wipes that are presaturated with isopropyl alcohol, the solvent of choice for its enhanced ability to clean multiple surfaces within a cleanroom. IPA’s purity and consistency make it an ideal solvent, especially when compared with denatured alcohol, which is often used in non-critical environments and contains up to 10% by volume of adulterating compounds. For more on IPA vs. denatured alcohol, please see our information sheet. And with any cleaner it’s essential to remember that disinfectant contact time – what the FDA terms ‘dwell time’ – is important, so be sure to follow any manufacturer’s guidelines about how long to leave the cleaner on a given surface.
In addition to selecting the correct cleaning consumables, operator training and adherence to strict standard operating procedures (SOPs) is the next line of defense against contamination. SOPs cover everything to do with operating the cleanroom environment – from personnel hygiene practices to allowable materials, to gowning procedures and general housekeeping. All critical environments should have them – do you? Devising and keeping your SOPs up-to-date is a major part of operating a safe and healthy cleanroom. But bear in mind that even the most rigorous SOPs are not worth the paper they’re written on if they’re not maintained and enforced. Berkshire recommends regular audits to ensure conformance to your SOPs – audits that are performed in person within the cleanroom itself. For a roadmap on how to ensure conformance by performing audits, please download our technical brief, ‘Auditing Conformance to Standard Operating Procedures.’
This might seem like a lot to absorb but in our industries – where we are creating products that directly impact public health and safety – no step goes too far, no warning is too strong. As we’ve seen in the last few years, FDA actions against compounding pharmacies that do not maintain a healthy, sterile environment in the production of their goods face stiff penalties. And that’s how it should be. Because, as the cases of NECC, of Franck’s Labs in Florida, or of Professional Arts Pharmacy in Maryland have so tragically demonstrated, lives are at stake and there are no second chances.
References:
- http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM514666.pdf
- http://the-difference-between.com/insanitary/unsanitary
- http://www.pressherald.com/2016/02/14/sullivan-harbor-farm-shut-down-for-seafood-safety-violations/
- See articles http://cleanroomnews.wpengine.com/2016/04/contamination-control-news-honey-not-sweet/, http://cleanroomnews.wpengine.com/2016/05/contamination-control-news-compounding-problem/, and http://cleanroomnews.wpengine.com/2016/09/when-is-a-pharmacy-not-pharmacy/
- http://www.newsweek.com/2015/04/24/inside-one-most-murderous-corporate-crimes-us-history-322665.html
- http://www.metrowestdailynews.com/news/20160625/co-founder-of-new-england-compounding-center-freed-from-home-detention
Additional references:
http://crimeblog.dallasnews.com/2016/02/texas-pharmacy-regulators-using-new-law-to-scrutinize-financial-ties-to-doctors.html/#more-110212
http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/PharmacyCompounding/ucm166743.htm



















I couldn’t refrain from commenting. Exceptionally well written!
Pingback: Regulatory Limits – What Is the Role of the FDA in International Trade? - Cleanroom News | Berkshire Corporation
Pingback: Exploring the Insanitary/Unsanitary Conundrum - Compounding Pharmacies - Berkshire Singapore
Pingback: Compounding Pharmacies: Exploring the Insanitary/Unsanitary Conundrum – USP 797 Compliance
Pingback: Compounding Pharmacies: Exploring the Insanitar...
“A big thank you for your blog article.Thanks Again. Will read on…”
Pingback: Should Cellphones Be Allowed In A Cleanroom? - Cleanroom News | Berkshire Corporation
Good post! I read your blog often and you always post excellent content. I posted this article on Facebook and my followers like it. Thanks for writing this!
Woah this blog is great i love reading your posts. Keep up the great work! You know, a lot of people are looking around for this information.
Great article. Thank you for your time in helping me learn about this.